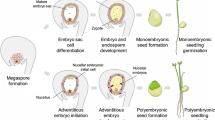
Abstract
Adventive polyembryony is a form of apomixis where sporophytic cells of the ovule directly give rise to embryos nourished by the endosperm of autonomous or sexual origin. Molecular basis of adventive embryony is poorly understood. This is the first report of transcriptome analysis of developing ovules during citrus nucellar embryo initiation. Suppression subtractive hybridization (SSH) and microarray were employed to find differentially expressed transcripts during early stages of seed development. Results of the two approaches were congruent for enriched GO terms (response to stress and signalling) and differential gene expression levels. SSH added 85 novel unigenes to the citrus EST database and revealed 443 new unigenes not represented in the Citrus Affymetrix array. Examination of the most-specific-GO-terms showed upregulation (123) and over-representation of transcripts related to abiotic stress (HSPs, HSF, pyruvate decarboxylase) and downregulation (92) of signalling-related (kinases and WRKYs) transcripts at the pre-anthesis stage of ovule development, when nucellar cells destined for embryogenesis begin to differentiate. Microarray and quantitative real-time polymerase chain reaction (qRT-PCR) analyses of genes present in the polyembryony locus identified significant differential expression of heat-shock-related genes and three novel ORFs (orf21, 22, and 25) between ovules of poly- and mono-embryonic varieties. qRT-PCR and microarray data confirmed the up- and downregulation of HSP and WRKY transcripts, respectively, in the pre-anthesis stage ovules of the polyembryonic variety. Based on differential gene expression pattern, a possible mechanistic sequence of polyembryony is presented.






Similar content being viewed by others
References
Altschul SF, Gish W, Miller W, Myres EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Angenent GC, Colombo L (1996) Molecular control of ovule development. Trends Plant Sci 1:228–232
Bicknell RA, Koltunow AM (2004) Understanding apomixis: recent advances and remaining conundrums. Plant Cell 16:S228–S245
Boutilier K, Offringa R, Sharma VK, Kieft H, Ouellet T, Zhang LM, Hattori J, Liu CM, van Lammeren AAM, Miki BLA, Custers JBM, Campagne MMV (2002) Ectopic expression of BABY BOOM triggers a conversion from vegetative to embryonic growth. Plant Cell 14:1737–1749
Brien TPO, Feder N, McCully ME (1964) Polychromatic staining of plant cell walls by toluidine blue O. Protoplasma 59:367–373
Cao W, Epstein C, Liu H, DeLoughery C, Ge N, Lin J, Diao R, Cao H, Long F, Zhang X, Chen Y, Wright PS, Busch S, Wenck M, Wong K, Saltzman AG, Tang Z, Liu L, Zilberstein A (2004) Comparing gene discovery from Affymetrix Gene Chip microarrays and Clontech PCR-select cDNA subtraction: a case study. BMC Genomics 5:26
Church M, Gilbert W (1984) Genomic sequencing. Proc Natl Acad Sci USA 81:1991–1995
Conesa A, Gotz S, Garcia-Gomez JM, Terol J, Talon M, Robles M (2005) Blast2GO: A universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21:3674–3676
Dean G, Cao YG, Xiang DQ, Provart NJ, Ramsay L, Ahad A, White R, Selvaraj G, Datla R, Haughn G (2011) Analysis of gene expression patterns during seed coat development in Arabidopsis. Mol Plant 4:1074–1091
deHoon MJ, Imoto S, Nolan J, Miyano S (2004) Open source clustering software. Bioinformatics 20:1453–1454
Despres B, Delseny M, Devic M (2001) Partial complementation of embryo defective mutations, a general strategy to elucidate gene function. Plant J 27:149–159
Esen A, Soost RK (1977) Adventive embryogenesis in citrus and its relation to pollination and fertilization. Am J Bot 64:607–614
Garcia R, Asins MJ, Forner J, Carbonell EA (1999) Genetic analysis of apomixis in Citrus and Poncirus by molecular markers. Theor Appl Genet 99:511–518
Garcia-Lor A, Garcia-Martinez JL, Perez-Amador MA (2012) Identification of ovule and seed genes from Citrus clementina. Tree Genet Genomes 8:227–235
Garcion C, Guilleminot J, Kroj T, Parcy F, Giraudat J, Devic M (2006) AKRP and EMB506 are two ankyrin repeat proteins essential for plastid differentiation and plant development in Arabidopsis. Plant J 48:895–906
Ge XX, Chai LJ, Liu Z, Wu XM, Deng XX, Guo WW (2012) Transcriptional profiling of genes involved in embryogenic, non-embryogenic calluses and somatic embryogenesis of Valencia sweet orange by SSH-based microarray. Planta 236:1107–1124
Hong QB, Xiang SQ, Chen KL, Chen LG (2001) Two complementary dominant genes controlling apomixis in genus Citrus and Poncirus. Acta Genet Sin 28:1062–1067
Jensen WA (1962) Botanical histochemistry—principles and practice. WH Freeman, San Francisco
Johnston AJ, Meier P, Gheyselinck J, Wuest SEJ, Federer M, Schlagenhauf E, Becker JD, Grossniklaus U (2007) Genetic subtraction profiling identifies genes essential for Arabidopsis reproduction and reveals interaction between the female gametophyte and the maternal sporophyte. Genome Biol 8:R204
Kepiro JL, Roose ML (2010) AFLP markers closely linked to a major gene essential for nucellar embryony (apomixis) in Citrus maxima × Poncirus trifoliata. Tree Genet Genomes 6:1–11
Kirik V, Kolle K, Misera S, Baumlein H (1998) Two novel MYB homologues with changed expression in late embryogenesis-defective Arabidopsis mutants. Plant Mol Biol 37:819–827
Kobayashi S, Ieda I, Nakantani M (1979) Studies on the nucellar embryogenesis in Citrus. Formation of the primordium cell of the nucellar embryo in the ovule of the flower bud, and its meristematic activity. J Jpn Soc Hort Sci 48:179–185
Koltunow AM (1993) Apomixis: embryo sacs and embryos formed without meiosis or fertilization in ovules. Plant Cell 5:1425–1437
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Lotan T, Ohto M, Yee KM, West MAL, Lo R, Kwong RW, Yamagishi K, Fischer RL, Goldberg RB, Harada JJ (1998) Arabidopsis LEAFY COTYLEDON1 is sufficient to induce embryo development in vegetative cells. Cell 93:1195–1205
Maraschin SF, de Priester W, Spaink HP, Wang M (2005) Androgenic switch: an example of plant embryogenesis from the male gametophyte perspective. J Exp Bot 56:1711–1726
Marco C, Roberta B (2006) BLAST Parser (the excel style viewer). BITS Conference (Bologna, Italy). Abstract no. 115
Mittler R, Finka A, Goloubinoff P (2012) How do plants feel the heat? Trend Biochem Sci 37:118–125
Mostafavi S, Ray D, Farley DW, Grouios C, Morris Q (2008) GeneMANIA: a real-time multiple association network integration algorithm for predicting gene function. Genome Biol 9:S4
Nakano M, Shimizu T, Fuji H, Shimada T, Endo T, Nesumi H, Kuniga T, Omura M (2008a) Marker enrichment and construction of haplotype-specific BAC contigs for the polyembryony region in Citrus. Breed Sci 58:375–383
Nakano M, Shimizu T, Kuniga T, Nesumi H, Omura M (2008b) Mapping and haplotyping of the flanking region of the polyembryony locus in Citrus unshiu Marcow. J Jpn Soc Hort Sci 77:109–114
Nakano M, Shimada T, Endo T, Fujii H, Nesumi H, Kita M, Ebina M, Shimizu T, Omura M (2012) Characterization of genomic sequence showing strong association with polyembryony among diverse Citrus species and cultivars, and its synteny with Vitis and Populus. Plant Sci 183:131–142
Nakano M, Kigoshi K, Shimizu T, Endo T, Shimada T, Fujii H, Omura M (2013) Characterization of genes associated with polyembryony and in vitro somatic embryogenesis in Citrus. Tree Genet Genomes 9:795–803
Pagnussat GC, Alandete-Saez M, Bowman JL, Sundaresan V (2009) Auxin-dependent patterning and gamete specification in the Arabidopsis female gametophyte. Science 324:1684–1689
Rose RJ, Nolan KE (2006) Genetic regulation of somatic embryogenesis with particular reference to Arabidopsis thaliana and Medicago truncatula. In Vitro Cell Dev Biol-Plant 42:473–481
Sánchez-León N, Arteaga-Vázquez M, Alvarez-Mejía C, Mendiola-Soto J, Durán-Figueroa N, Rodríguez-Leal D, Rodríguez-Arévalo I, García-Campayo V, García-Aguilar M, Olmedo-Monfil V, Arteaga-Sánchez M, de la Vega OM, Nobuta K, Vemaraju K, Meyers BC, Vielle-Calzada JP (2012) Transcriptional analysis of the Arabidopsis ovule by massively parallel signature sequencing. J Exp Bot 63:3829–3842
Schmidt EDL, Guzzo F, Toonen MAJ, de Vries S (1997) A leucine rich repeat containing receptor-like kinase marks somatic plant cells competent to form embryos. Development 124:2049–2062
Schmidt A, Wuest SE, Vijverberg K, Baroux C, Kleen D, Grossniklaus U (2011) Transcriptome analysis of the Arabidopsis megaspore mother cell uncovers the importance of RNA helicases for plant germline development. PLoS Biol 9:e1001155
Steffen JG, Kang IH, Macfarlane J, Drews GN (2007) Identification of genes expressed in the Arabidopsis female gametophyte. Plant J 51:281–292
Talon M, Gmitter FG Jr (2008) Citrus genomics. Int J Plant Genomics 528361
Wu KL, Guo ZJ, Wang HH, Li J (2005) The WRKY family of transcription factors in rice and Arabidopsis and their origins. DNA Res 12:9–26
Xu Q, Chen LL, Ruan X, Chen D, Zhu A, Chen C, Bertrand D, Jiao WB, Hao BH, Lyon MP, Chen J, Gao S, Xing F, Lan H, Chang JW, Ge X, Lei Y, Hu Q, Miao Y, Wang L, Xiao S, Biswas MK, Zeng W, Guo F, Cao H, Yang X, Xu XW, Cheng YJ, Xu J, Liu JH, Luo OJ, Tang Z, Guo WW, Kuang H, Zhang HY, Roose ML, Nagarajan N, Deng XX, Ruan Y (2013) The draft genome of sweet orange (Citrus sinensis). Nat Genet 45:59–66
Yang H (2006) EST generation and analyses towards identifying female gametophyte-specific genes in Zea mays L. Planta 224:1004–1014
Yang X, Zang X (2010) Regulation of somatic embryogenesis in higher plants. Crit Rev Plant Sci 29:36–57
Yang WC, Shi DQ, Chen YH (2010) Female gametophyte development in flowering plants. Annu Rev Plant Biol 61:89–108
Yu HJ, Hogan P, Sundaresan V (2005) Analysis of the female gametophyte transcriptome of Arabidopsis by comparative expression profiling. Plant Physiol 139:1853–1869
Zavattieri MA, Frederico AM, Lima M, Sabino R, Arnholdt-Schmitt B (2010) Induction of somatic embryogenesis as an example of stress-related plant reactions. Electron J Biotechnol 13:1–9
Zhu M, Zhao S (2007) Candidate gene identification approach: progress and challenges. Int J Biol Sci 3:420–427
Acknowledgments
We thank J. S. Josan, Punjab Agricultural University Regional Station, Abohar, Punjab, for the supply of citrus materials used in this study. We acknowledge the critical comments of A. Bandyopadhyay. We thank P. A. Kumar and Neha Jain, NRCPB, for assisting in microarray data analysis. This project was funded by a World Bank supported National Agricultural Innovation Project of the Indian Council of Agricultural Research, New Delhi, India.
Data archiving statement
The SSH ESTs generated were deposited in GenBank under accession numbers HO046617-HO048242 and JZ119940-JZ120395. Microarray data and associated experimental details of this work were deposited in the Gene Expression Omnibus (http://www.ncbi.nlm.nih.gov/geo/) under the accession number GSE36188 (GSM883400 to GSM883420).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by W.-W. Guo
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1 (download DOC )
(DOC 1176 kb)
ESM 2 (download DOC )
(DOC 2198 kb)
Rights and permissions
About this article
Cite this article
Kumar, V., Malik, S.K., Pal, D. et al. Comparative transcriptome analysis of ovules reveals stress related genes associated with nucellar polyembryony in citrus. Tree Genetics & Genomes 10, 449–464 (2014). https://doi.org/10.1007/s11295-013-0690-0
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1007/s11295-013-0690-0